The hypertension market is no stranger to new entrants, with therapies ranging from calcium channel blockers and angiotensin II receptor blockers (ARBs) to angiotensin-converting enzyme (ACE) inhibitors and beta blockers. As a mature market, generics are widely available, and many of the more recently approved therapies are combinations of existing agents or different formulations of FDA-approved treatments.
So in a market with long-established therapies and plenty of branded and generic competition, do new market entrants stand a chance? And what does the space look like for established brands vs. new ones? MMIT’s market access team did a deep dive into the current state of the hypertension market. Here’s what manufacturers need to know.
Market Overview
The hypertension space has seen plenty of recent activity. On March 19, 2024, the FDA approved Idorsia Pharmaceuticals U.S. Inc.’s Tryvio (aprocitentan), in combination with other antihypertensive drugs, to lower blood pressure in adults who are not adequately controlled on other drugs. The endothelin receptor antagonist is the first oral anti-hypertensive treatment approved in nearly 40 years that works via a new therapeutic pathway. The agent will launch in the second half of 2024.
On Feb. 24, 2022, the FDA approved CMP Pharma Inc.’s Norliqva (amlodipine besylate), a long-acting calcium channel blocker, to lower blood pressure in adults and children at least 6 years old. The agent was the first liquid solution of amlodipine that the FDA approved to treat hypertension in people 6 years of age and older, making it well suited for people who have difficulty swallowing or the inability to swallow.
The Hypertension Patient Journey
Most patients start on one of the first-line agents, and then a second is added if blood pressure goals aren’t reached. Combination agents and once-daily dosing are often used for their convenience and ability to promote compliance.
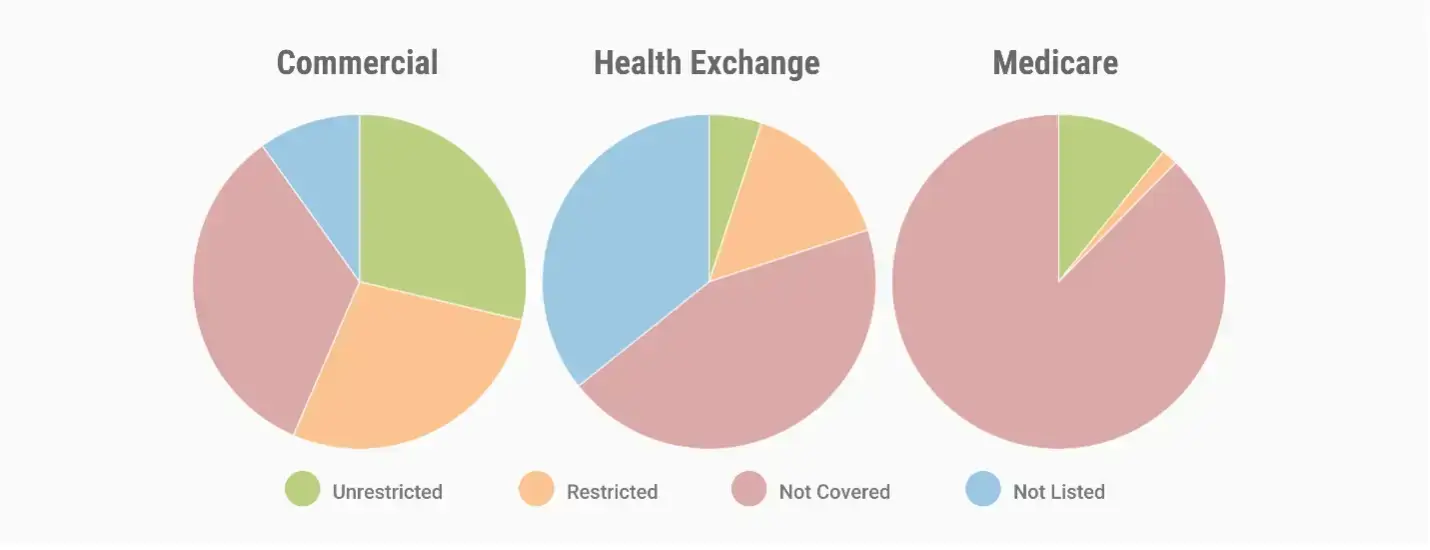
How Are Hypertension Drugs Covered?
An MMIT Analytics market access review for hypertension treatments shows that under the pharmacy benefit, about 38% of lives across all lines of business are covered for at least one treatment with utilization management (UM) restrictions.
For about 68% of the covered lives, payer pharmacy benefit formularies do not require step therapy. Of the lives that are subject to required step therapy, approximately 30% are required to complete multiple steps. Exceptions to trial and failure include scenarios in which a member is unable to swallow tablets or capsules, as well as scenarios in which a member’s condition is clinically stable and switching therapies would cause deterioration.
Around 30% of payer-controlled pharmacy benefit covered lives require prior authorization, with about 37% of those lives covered by policies that are restrictive as compared with a product’s FDA-approved label.
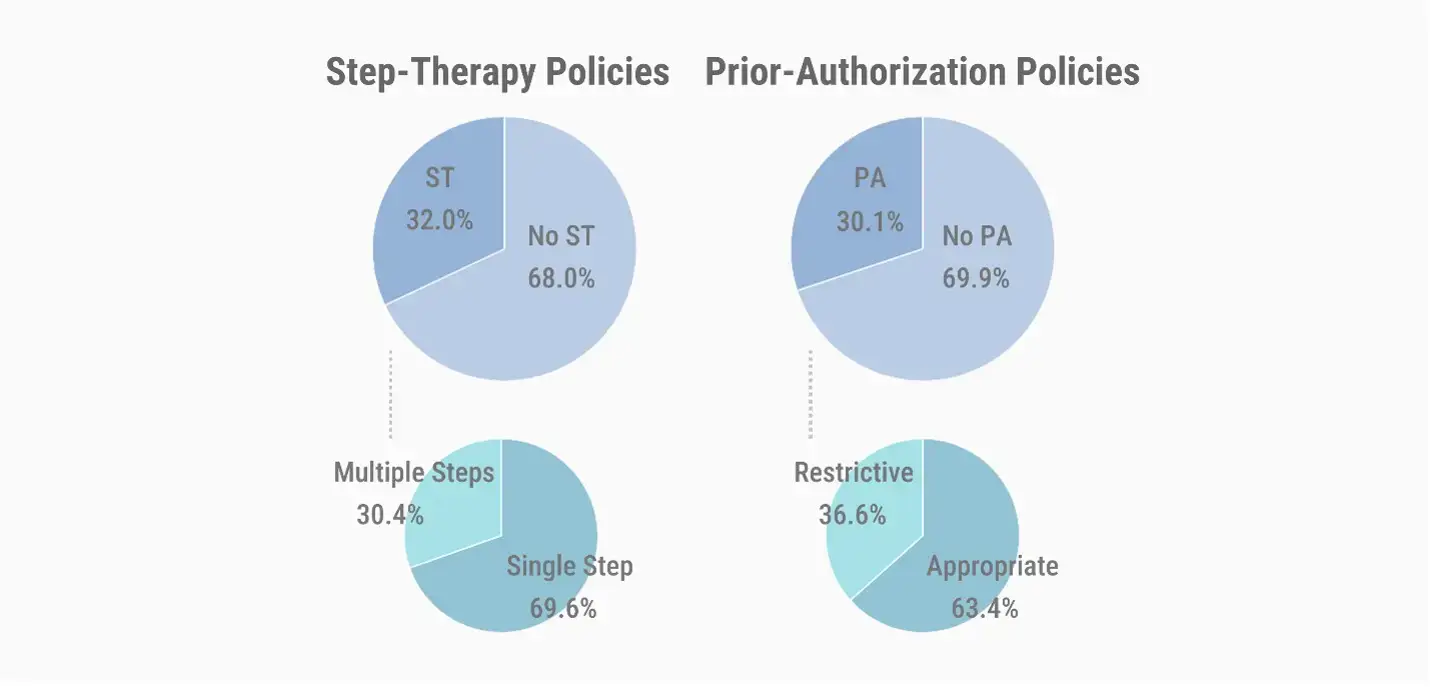
Around 10% of payer-controlled pharmacy benefit covered lives require quantity limits, as compared with 43% of covered lives that do not have quantity limits on the agents.
The Bottom Line
In general, coverage for hypertension therapies is quite similar across the board, without much differentiation. Few new or novel hypertension therapies have been developed in recent years.
Some companies have distinguished their products by offering formulations that are easy to swallow for people who have difficulty swallowing pills, but that likely isn’t enough, as many generics can be crushed into liquid or applesauce for easier consumption.
In this mature market with widely available generics, it’s rare for a brand-name hypertension drug to be preferred on formulary. While this therapeutic class doesn’t have a lot of specialist requirements, new market entrants will find gaining a foothold to be challenging. Unless a product comes to market generically priced, it will likely struggle to gain market share.
Looking to evaluate market access for your brand and competitors? MMIT’s Analytics solution delivers the most comprehensive formulary, medical policy and restriction information available.






